
The final aim of HyMAP is to develop hybrid materials for artificial photosynthesis, which encompasses mainly two processes: CO2 conversion and hydrogen production. We test our organic, inorganic and hybrid materials in these processes using laboratory scale reactors. We do not, however, overlook process scalability and, with that in mind, we seek for new preparation procedures for highly active, scalable and versatile photoactive materials. Our engineering point of view is also focused on the design of efficient reactors and devices to carry out the pursued process of sun-driving the conversion of natural feedstocks into fuels, chemicals and energy vectors.
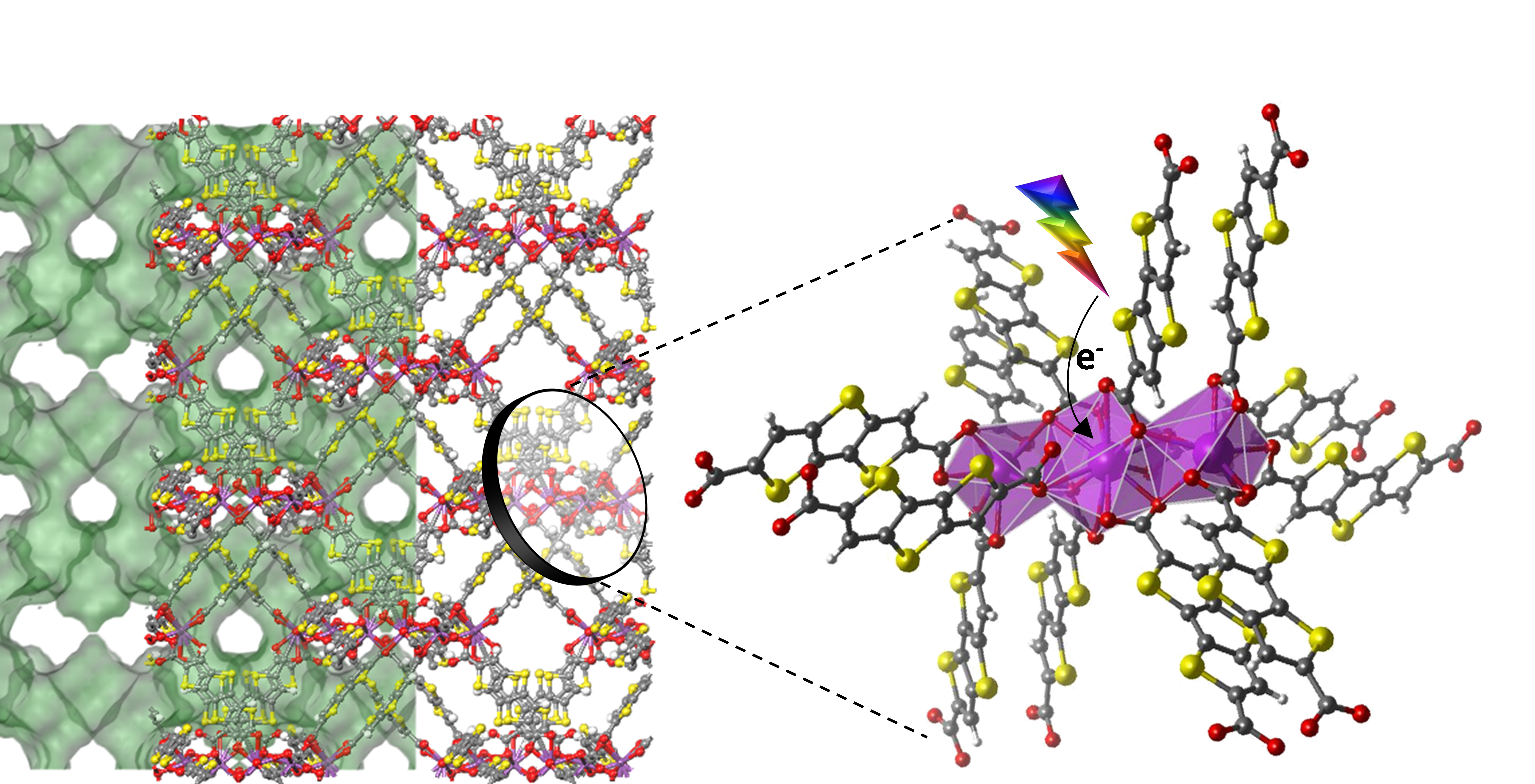
Metal-Organic Frameworks (MOFs) are porous networks generates by the combination of organic ligands and metal ions thought coordination bonds. These novel hybrid materials have attracted much attention during the recent years due their tuneable structures and their great variety of properties such as, gas and chemical storage, catalysis, ion exchange, and sensing, among them our group is interested especially in their use as catalyst for the production of clean energy.
One of our objectives the design and synthesis of new structures as photocatalyst to solar fuel production. Basically, this seeking consist in the developing of organic ligands that allow light harvesting and electron transport to selected metal active sites to transform CO2 and H2O in fuels and chemicals via Artificial photosynthesis using sunlight as renewable energy source.

We use advanced photoelectron spectroscopy techniques such as XPS, NEXAFS and RPES to probe the electronic state of the main actors involved in the photocatalytic process at operando conditions, i.e. under UV-irradiation and CO2/H2O atmosphere. XPS and NEXAFS help us to detect changes in the oxidation state of the catalyst and to identify reaction intermediates and products at its surface. XPS and RPES are used to map the valence band of the photocatalyst and identify atomic contributions. The core-hole-clock technique (from XAS/RPES or XAS/RAES data) can be used to measure charge transfer times during the photocatalytic process.
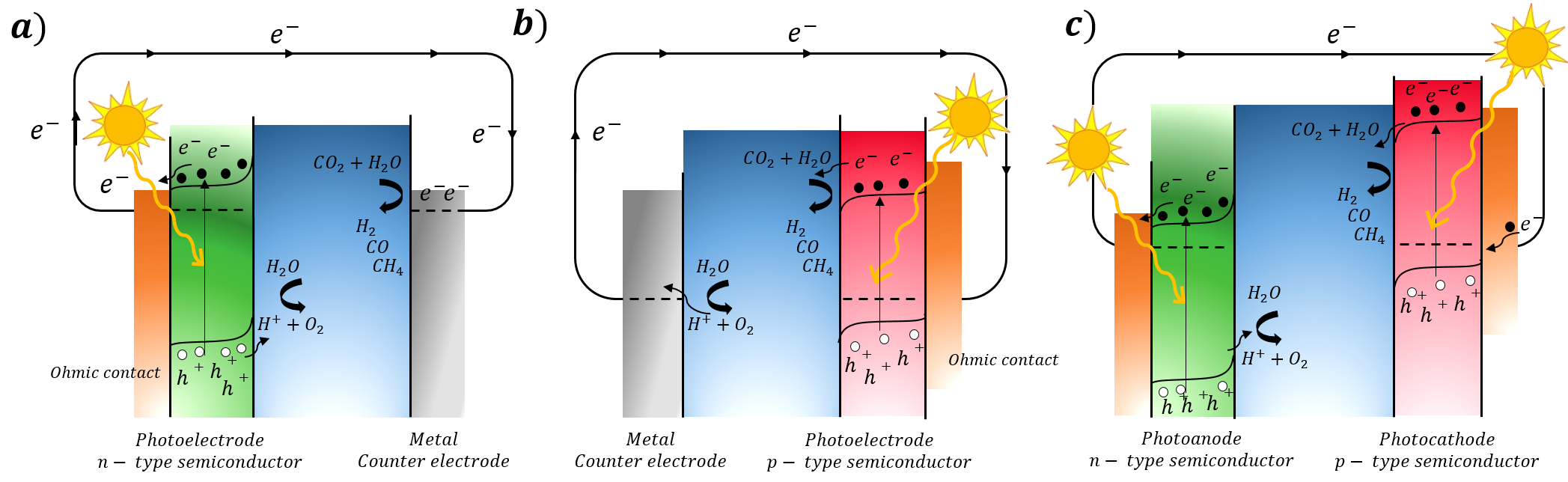
Solar energy can be transformed into chemical energy by means of photocatalytic reactions and photoelectrochemical cells (PEC), which can be call artificial photosynthesis. In this way, semiconductor materials have demonstrated their ability to use it as photocatalyst and split water into separated O2 and H2 gases, or to reduce CO2. In this lab, we designed and prepare different semiconductors with specific optical and electronic properties and use it to build hybrid and smart materials. For that, we carry out inorganic (metal oxides) and organic (conjugated organic polymers with high-conductivity) synthesis.
To test this materials, we use different set-ups: a conventional and a tandem cell. A conventional PEC consists of two indispensable parts: efficient light collectors as working electrodes (anode, figure a, or cathode, figure b, compartment), and a metal as counter electrode. A promising approach is the combination of stable and efficient materials in a tandem photoelectrochemical device (figure c). Our photoelectrochemistry line research is focused on the designed and use of these smarts and hybrid materials to be used as photoelectrodes (photoanodes and photocathodes) in water splitting and CO2 reduction reactions.